This article was co-authored by Bess Ruff, MA. Bess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
There are 8 references cited in this article, which can be found at the bottom of the page.
This article has been viewed 1,149,837 times.
Although all atoms of the same element contain the same number of protons, their number of neutrons can vary. Knowing how many neutrons are in a particular atom can help you determine if it's a regular atom of that element or an isotope, which will have either extra or fewer neutrons.[1] Determining the number of neutrons in an atom is fairly simple and doesn’t even require any experimentation. To calculate the number of neutrons in a regular atom or an isotope, all you need to do is follow these instructions with a periodic table in hand.
Steps
Finding the Number of Neutrons in a Regular Atom
-
1Locate the element on the periodic table. For this example, we’ll look at osmium (Os), which is in the sixth row down.[2]
-
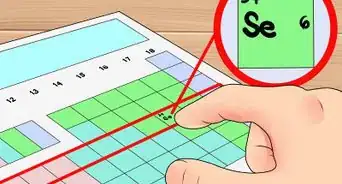
2Find the element’s atomic number. This tends to be the most visible number pertaining to a given element and usually sits above the element symbol, either in the middle of the box or in the upper left corner. (On the chart we're using, in fact, no other numbers are listed.) The atomic number is the number of protons in a single atom of that element.[3] Os is number 76, meaning one atom of osmium has 76 protons.
- The proton number never changes in an element; it's basically what makes that element that element.[4]
Advertisement -
3Find the element’s atomic weight. This number is usually found beneath the atomic symbol. Note that the chart in this example is based solely on atomic number and doesn’t list the atomic weight. This won’t always usually be the case. Osmium has an atomic weight of 190.23.[5]
-
4Round off the atomic weight to the nearest whole number to find the atomic mass.[6] In our example, 190.23 would be rounded to 190, resulting in an atomic mass of 190 for osmium.
- The atomic weight is an average of the isotopes of the element, so that's why it's not usually a whole number.[7]
-
5Subtract the atomic number from the atomic mass. Since the vast majority of an atom’s mass is made up of its protons and neutrons, subtracting the number of protons (i.e. the atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. In our example, this is: 190 (atomic weight) – 76 (number of protons) = 114 (number of neutrons).[8]
-
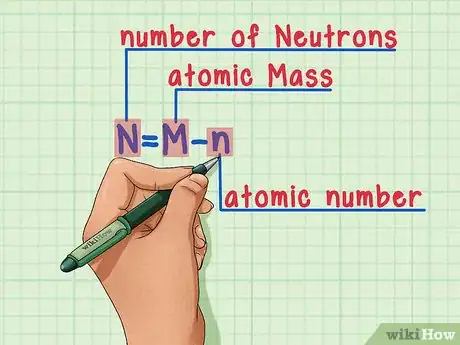
6Remember the formula. To find the number of neutrons in the future, simply use this formula:[9]
-
N = M – n
- N = number of Neutrons
- M = atomic Mass
- n = atomic number
-
N = M – n
Finding the Number of Neutrons in an Isotope
-
1Locate the element on the periodic table. As an example, we’ll look at the carbon-14 isotope. Since the non-isotopic form of carbon-14 is simply carbon (C), find carbon on the periodic table (in the second row down).[10]
-
2Find the element’s atomic number. This tends to be the most visible number pertaining to a given element and usually sits above the element symbol. (On our example chart, in fact, no other numbers are listed.) The atomic number is the number of protons in a single atom of that element.[11] C is number 6, meaning one atom of carbon has 6 protons.
-
3Find the atomic mass. This is incredibly easy with isotopes, as they are named according to their atomic mass. Carbon-14, for example, has an atomic mass of 14. Once you find the atomic mass of the isotope, the process is the same as it is for finding the number of neutrons in a regular atom.[12]
-
4Subtract the atomic number from the atomic mass. Since the vast majority of an atom’s mass is found its protons and neutrons, subtracting the number of protons (i.e. the atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. In our example, this is: 14 (atomic mass) – 6 (number of protons) = 8 (number of neutrons).
-
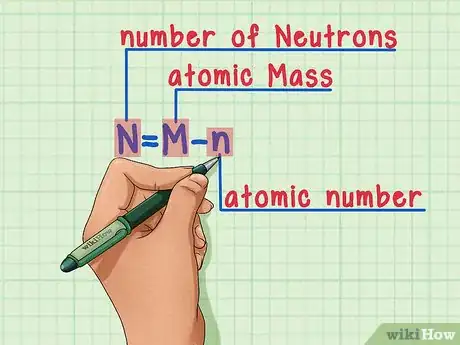
5Remember the formula. To find the number of neutrons in the future, simply use this formula:[13]
-
N = M – n
- N = number of Neutrons
- M = atomic Mass
- n = atomic number
-
N = M – n
Expert Q&A
Did you know you can get expert answers for this article?
Unlock expert answers by supporting wikiHow
-
QuestionHow do you find the number of electrons, neutrons and protons?
 Bess Ruff, MABess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
Bess Ruff, MABess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
Environmental Scientist
-
QuestionHow many neutrons are in oxygen?
 Community AnswerAtomic mass minus the atomic number. Atomic mass of Oxygen is 16, and the atomic number is 8. 16 - 8 = 8.
Community AnswerAtomic mass minus the atomic number. Atomic mass of Oxygen is 16, and the atomic number is 8. 16 - 8 = 8. -
QuestionWhere can I locate the atomic mass in a modern periodic table?
 Community AnswerThe modern periodic table has the atomic number on the top of each element's symbol and the atomic mass right below the symbol.
Community AnswerThe modern periodic table has the atomic number on the top of each element's symbol and the atomic mass right below the symbol.
References
- ↑ https://chem.libretexts.org/LibreTexts/College_of_Marin/Marin%3A_CHEM_114_-_Introductory_Chemistry_(Daubenmire)/04%3A_Atoms_and_Elements/4.8%3A_Isotopes%3A_When_the_Number_of_Neutrons_Varies
- ↑ https://periodic.lanl.gov/index.shtml
- ↑ http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch6/index.php
- ↑ https://www.bbc.com/bitesize/guides/z3sg2nb/revision/4
- ↑ https://www.khanacademy.org/science/biology/chemistry--of-life/elements-and-atoms/a/atomic-number-atomic-mass-and-isotopes-article
- ↑ https://chem.libretexts.org/Courses/Furman_University/CHM101%3A_Chemistry_and_Global_Awareness_(Gordon)/03%3A_Atoms_and_the_Periodic_Table/3.04%3A_Atomic_Mass_and_Atomic_Number
- ↑ http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch3/massmol.html
- ↑ https://chem.libretexts.org/Courses/Furman_University/CHM101%3A_Chemistry_and_Global_Awareness_(Gordon)/03%3A_Atoms_and_the_Periodic_Table/3.04%3A_Atomic_Mass_and_Atomic_Number
- ↑ https://chem.libretexts.org/Courses/Furman_University/CHM101%3A_Chemistry_and_Global_Awareness_(Gordon)/03%3A_Atoms_and_the_Periodic_Table/3.04%3A_Atomic_Mass_and_Atomic_Number
- ↑ https://periodic.lanl.gov/index.shtml
- ↑ https://www.bbc.com/bitesize/guides/z3sg2nb/revision/4
- ↑ https://www.khanacademy.org/science/biology/chemistry--of-life/elements-and-atoms/a/atomic-number-atomic-mass-and-isotopes-article
- ↑ https://chem.libretexts.org/Courses/Furman_University/CHM101%3A_Chemistry_and_Global_Awareness_(Gordon)/03%3A_Atoms_and_the_Periodic_Table/3.04%3A_Atomic_Mass_and_Atomic_Number
- ↑ https://www.khanacademy.org/science/biology/chemistry--of-life/elements-and-atoms/a/atomic-number-atomic-mass-and-isotopes-article
- Interactive periodic table
About This Article
To find the number of neutrons in an atom, you just need a periodic table that lists the atomic number as well as the atomic weight of each element. First, locate the elemental symbol for your atom on the periodic table. Then, find the atomic number for the element. It’s usually located somewhere above the element symbol. This number represents the number of protons in the atom. For instance, the atomic number of niobium (Nb) is 41, meaning that a niobium atom has 41 protons. Next, find the atomic weight of the element, which is usually underneath the element symbol. Round up the atomic weight to the nearest whole number. Niobium has an atomic weight of 92.906, so you would round it up to 93. Finally, subtract the number of protons from the rounded up atomic weight to find the number of neutrons in the atom. In the case of niobium, 93 minus 41 is 52, which means that a niobium atom has 52 neutrons. To find out how to calculate the number of neutrons in an isotope, read on!