Proteases are enzymes that have the ability to cut proteins into peptides. They are used by some viruses (e.g., HIV) to cleave precursor long protein chains into individual proteins. This allows the completion of the assembly step in the viral life cycle where the proteins and the viral RNA come together to form virion particles ready to exit the cell.
The design of protease inhibitors, that could be used to battle HIV, started soon after the discovery of the virus. The first approved protease inhibitor drug was released on the market in 1995, only 10 years after the discovery of HIV. These drugs are an inseparable part of an HIV therapy. Natural protease inhibitors are found in Shiitake mushrooms. The experimental protease inhibitor drugs Zmapp and Brincidofovir are currently being tested to treat the ebola virus disease.
Protease inhibitors are short peptide-like molecules that are competitive inhibitors of the enzyme. Instead of -NH-CO- peptide link, they contain -(CH2-CH(OH)-). When such a peptide gets into the enzyme active site, the protease is unable to cut the linkage and gets inactivated. This leads to a lack of cleavage of the polypeptide chains of two crucial viral proteins, Gag and Pol, which are essential structural and enzymatic proteins of HIV. Their absence blocks the formation of mature virion particles.
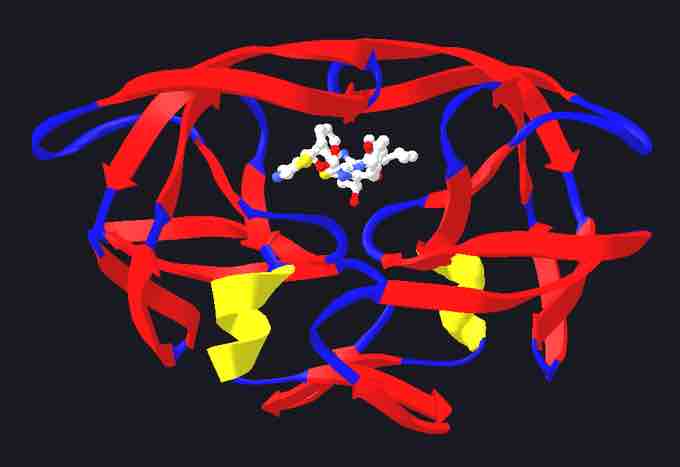
HIV protease with bound protease inhibitor
The drug is ritonavir depicted here with a white molecule in the middle of the enzyme structure.
Saquinavir is the first clinically used peptide-like inhibitor. Some protease inhibitors do not mimic peptides in their structure. One such drug is Nelfinavir. In general, protease inhibitors exhibit the unusual side effect of fat storage in non-typical organs and tissues. The reasons for this are still unclear.
Mutations in the enzyme active site and other sites, which cause conformational changes, can cause resistance. Quite often one mutation can lead to resistance to many different drugs simultaneously since they all share the same mode of action. This is called cross-resistance. It is one of the major drawbacks of protease inhibitors therapy.